- Internal Energy
Internal energy is all the mechanical energy in all the components of a
system. For example, in a monoatomic gas it might be the sum of the
kinetic energies of all the gas atoms. In a solid it might be the sum
of the kinetic and potential energies of all the particles that make up
the solid.
- Heat
Heat is a bit more complicated. It is internal energy as well, but it
is internal energy that is transferred into or out of a given
system. Furthermore, it is in some fundamental sense ``disorganized''
internal energy - energy with no particular organization, random
energy. Heat flows into or out of a system in response to a temperature
difference, always flowing from hotter temperature regions (cooling
them) to cooler ones (warming them).
Common units of heat include the ever-popular Joule and the calorie (the heat required to raise the temperature of 1 gram of water
at 14.5 C to 15.5
C to 15.5 C. Note that 1 cal = 4.186 J. Less
common and more esoteric ones like the British Thermal Unit (BTU) and
erg will be mostly ignored in this course; BTUs raise the temperature of
one pound of water by one degree Fahrenheit, for example. Ugly.
C. Note that 1 cal = 4.186 J. Less
common and more esoteric ones like the British Thermal Unit (BTU) and
erg will be mostly ignored in this course; BTUs raise the temperature of
one pound of water by one degree Fahrenheit, for example. Ugly.
- Heat Capacity
If one adds heat to an object, its temperature usually increases
(exceptions include at a state boundary, for example when a liquid
boils). In many cases the temperature change is linear in the
amount of heat added. We define the heat capacity  of an object
from the relation:
of an object
from the relation:
 |
(219) |
where  is the heat that flows into a system to increase its
temperature by
is the heat that flows into a system to increase its
temperature by  . Many substances have a known heat capacity
per unit mass. This permits us to also write:
. Many substances have a known heat capacity
per unit mass. This permits us to also write:
 |
(220) |
where  is the specific heat of a substance. The specific heat
of liquid water is approximately:
is the specific heat of a substance. The specific heat
of liquid water is approximately:
 |
(221) |
(as one might guess from the definition of the calorie above).
- Latent Heat
As noted above, there are particular times when one can add heat to a
system and not change its temperature. One such time is when the system
is changing state from/to solid to/from liquid, or from/to liquid
to/from gas. At those times, one adds (or removes) heat when the system
is at fixed temperature until the state change is complete. The
specific heat may well change across phase boundaries. There are two
trivial equations to learn:
where  is the latent heat of fusion and
is the latent heat of fusion and  is the latent heat of vaporization. Two important numbers to keep in mind are
is the latent heat of vaporization. Two important numbers to keep in mind are
 kJ/kg, and
kJ/kg, and
 kJ/kg. Note the high
value of the latter - the reason that ``steam burns worse than water''.
kJ/kg. Note the high
value of the latter - the reason that ``steam burns worse than water''.
- Work Done by a Gas
 |
(224) |
This is the area under the  curve, suggesting that we draw lots of
state diagrams on a
curve, suggesting that we draw lots of
state diagrams on a 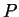 and
and  coordinate system. Both heat transfer
and word depend on the path a gas takes
coordinate system. Both heat transfer
and word depend on the path a gas takes  moving from one
pressure and volume to another.
moving from one
pressure and volume to another.
- The First Law of Thermodynamics
 |
(225) |
In words, this is that the change in total mechanical energy of a system
is equal to heat put into the system plus the work done on the
system (which is minus the work done by the system, hence the
minus above).
This is just, at long last, the fully generalized law of
conservation of energy. All the cases where mechanical energy was not
conserved in previous chapters because of nonconservative forces, the
missing energy appeared as heat, energy that naturally flows from
hotter systems to cooler ones.
- Cyclic Processes
Most of what we study in these final sections will lead us to an
understanding of simple heat engines based on gas expanding in a
cylinder and doing work against a piston. In order to build a true
engine, the engine has to go around in a repetitive cycle. This
cycle typically is represented by a closed loop on a state e.g.
 curve. A direct consequence of the 1st law is that the net work
done by the system per cycle is the area inside the loop of the
curve. A direct consequence of the 1st law is that the net work
done by the system per cycle is the area inside the loop of the  diagram. Since the internal energy is the same at the beginning and
the end of the cycle, it also tells us that:
diagram. Since the internal energy is the same at the beginning and
the end of the cycle, it also tells us that:
 |
(226) |
the heat that flows into the system per cycle must exactly equal the
work done by the system per cycle.
- Adiabatic Processes
are processes (
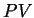 curves) such that no heat enters or leaves an
(insulated) system.
curves) such that no heat enters or leaves an
(insulated) system.
- Isothermal Processes
are processes where the temperature
 of the system remains constant.
of the system remains constant.
- Isobaric Processes
are processes that occur at constant pressure.
- Isovolumetric Processses
are processes that occur at constant volume.
- Work done by an Ideal Gas: Recall,
 |
(227) |
where  is the number of gas atoms or molecules. Isothermal work at
(fixed) temperature
is the number of gas atoms or molecules. Isothermal work at
(fixed) temperature  is thus:
is thus:
Isobaric work is trivial.  is a constant, so
is a constant, so
 |
(230) |
Adiabatic work is a bit tricky and depends on some of the internal
properties of the gas (for example, whether it is mono- or diatomic).
We'll examine this in the next section.
![]() C to 15.5
C to 15.5![]() C. Note that 1 cal = 4.186 J. Less
common and more esoteric ones like the British Thermal Unit (BTU) and
erg will be mostly ignored in this course; BTUs raise the temperature of
one pound of water by one degree Fahrenheit, for example. Ugly.
C. Note that 1 cal = 4.186 J. Less
common and more esoteric ones like the British Thermal Unit (BTU) and
erg will be mostly ignored in this course; BTUs raise the temperature of
one pound of water by one degree Fahrenheit, for example. Ugly.
![]() of an object
from the relation:
of an object
from the relation:


![]() is a constant, so
is a constant, so